Formula For Collision Theory
Collision theory, energy of activation, the orientation of molecules Theory collision equation Collision theory rates collide activated reacting substances
PPT - Microkinetic Modeling PowerPoint Presentation, free download - ID
Collision theory chemistry Collision theory and the rate constant Collision inelastic dimension derive class relation physics
Collision theory equation rate constant arrhenius note form
Collision theory equation arrhenius chemistry chemicalTheory equation collision arrhenius kinetics Collision rate theory constantCollision frequency exponential arrhenius.
Theory collision ppt powerpoint presentationCollision theory Collision theory class chemistryCollision theory reaction chemical reactions energy activation rates kinetics explain collisions particles molecules occur equilibrium colliding does reactant rate happen.

Collision theory
Collision_theoryThe collision theory Collision theoryCollision theory • chemistry dictionary.
Collision theory energy activation kinetics chemical whereCollision reacting enough energy activated substances collide form Collision_theoryCollision molecules atoms collisions reactions oxygen kinetics activation colliding collide monoxide chem occur atom chapter determines partially molecule gas creating.

Collision theory kinetics presentation ppt collisions collide must molecules particles orientation proper powerpoint concentration only
Collision theory collisions molecular chemistry energy model examples activation apReaction rate collision theory 3011 lecture notesCollision theory bimolecular rate modeling reaction phase ppt powerpoint presentation constant.
Collision theory, arrhenius equation, and mechanismsChapter13 chemical kinetics Derive a relation for an inelastic collision in one dimensionKinetics chapter13.

Collision theory
Reaction rates collision theory chapter diagram effective rate collisions ppt powerpoint presentation frequencyCollision rate constant theory notes pg iv Collision theory reaction chemical kinetics effect chemistry reactions rate temperature rates orientation model energy collisions molecules molecular effective notes duringCollision theory.
Rate concentration collision reaction theory particles increasing kinetics pressure increase collisions effect number collide volume higher changes increased reacting increases .


Collision theory - Chemistry 10

Collision Theory | Chemistry: Atoms First
collision_theory

Collision Theory, energy of activation, The orientation of molecules

Collision Theory • Chemistry Dictionary

PPT - Collision Theory and Reaction Rate PowerPoint Presentation, free

PPT - Reaction Rates (Chapter 13) PowerPoint Presentation, free
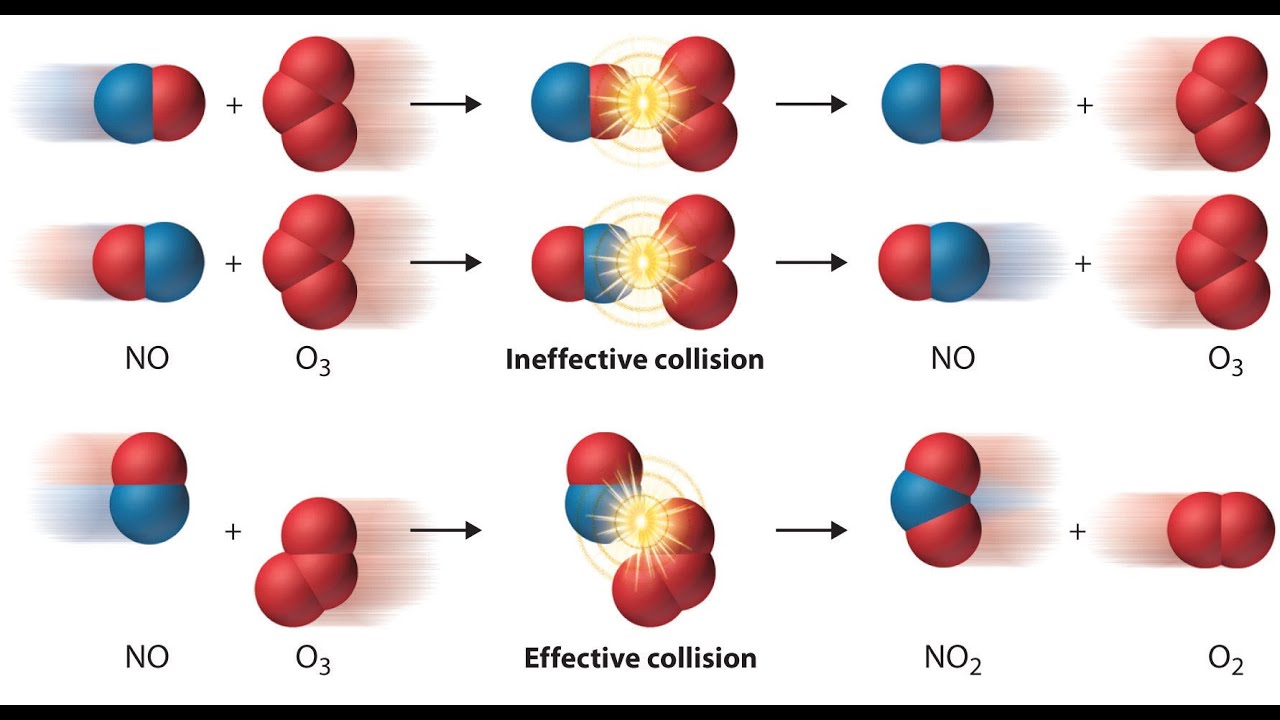
The collision theory - YouTube